 
She was the first to realize that Hahn's barium and other lighter products from the neutron bombardment experiments were coming from the fission of U-235. Upon Hitler's invasion of Austria, she had been forced to flee to Sweden where she and Otto Frisch, her nephew, continued to work on the neutron bombardment problem. When they finally published the results in 1939, they came to the attention of Lise Meitner, an Austrian-born physicist who had worked with Hahn on his nuclear experiments. When they finally identified one of the products as Barium-141, they were reluctant to publish the finding because it was so unexpected. Rather than the heavy elements they expected, they got several unidentified products. In the 1930s, German physicists/chemists Otto Hahn and Fritz Strassman attempted to create transuranic elements by bombarding uranium with neutrons. The only other isotope which is known to undergo fission upon slow-neutron bombardment is uranium-233. Thorium-232 is fissionable, so could conceivably be used as a nuclear fuel. Some of the nuclear reactors at Hanford, Washington and the Savannah-River Plant (SC) are designed for the production of bomb-grade plutonium-239. U-238 has a small probability for spontaneous fission and also a small probability of fission when bombarded with fast neutrons, but it is not useful as a nuclear fuel source. Uranium-238, which makes up 99.3% of natural uranium, is not fissionable by slow neutrons. Plutonium-239 can be produced by " breeding" it from uranium-238. Plutonium-239 is also fissionable by bombardment with slow neutrons, and both it and uranium-235 have been used to make nuclear fission bombs. While uranium-235 is the naturally occuring fissionable isotope, there are other isotopes which can be induced to fission by neutron bombardment. Even with the necessity of enrichment, it still takes only about 3 kg of natural uranium to supply the energy needs of one American for a year. After reduction, the uranium must go through an isotope enrichment process. Uranium is found as uranium oxide which when purified has a rich yellow color and is called "yellowcake". For light-water reactors, the fuel must be enriched to 2.5-3.5% U-235. 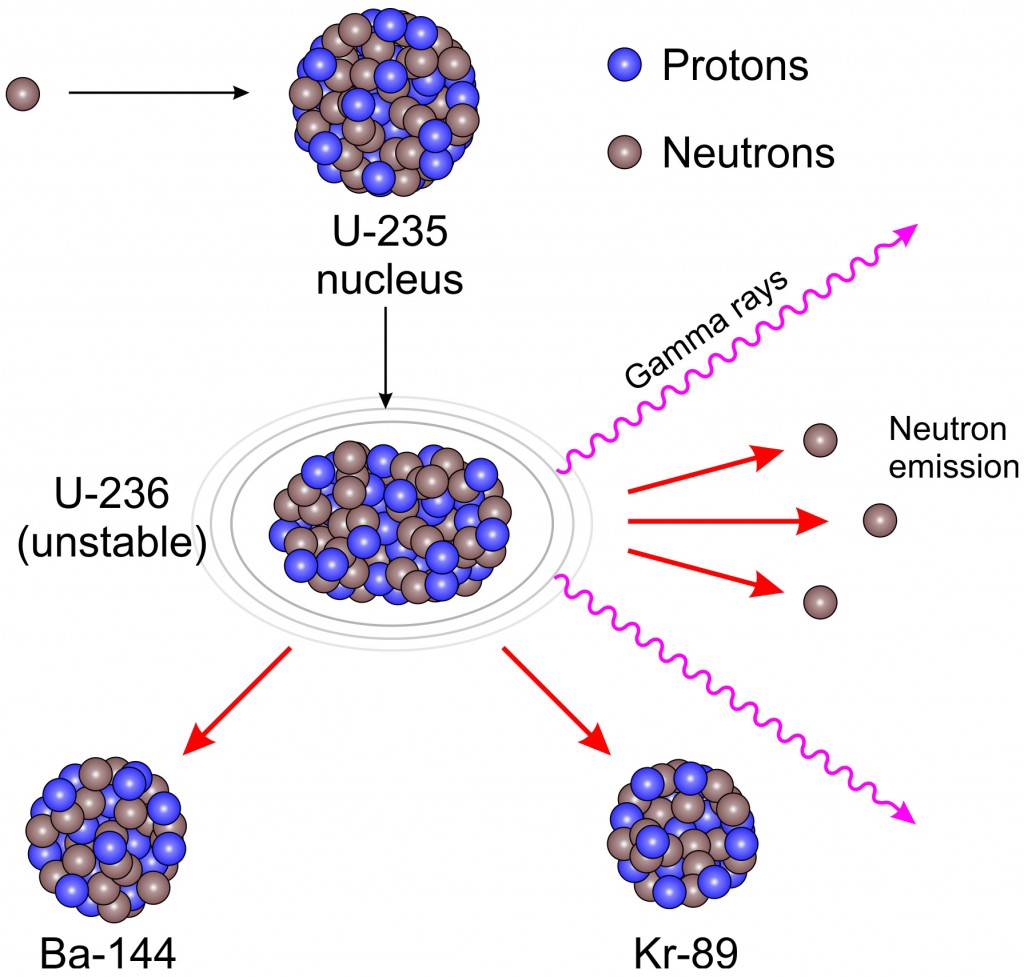
style light-water reactors, although it is used in Canadian CANDU reactors. 
The 0.72% U-235 is not sufficient to produce a self-sustaining critical chain reaction in U.S. Natural uranium is composed of 0.72% U-235 (the fissionable isotope), 99.27% U-238, and a trace quantity 0.0055% U-234. A single fission event can yield over 200 million times the energy of the neutron which triggered it! A fast neutron will not be captured, so neutrons must be slowed down by moderation to increase their capture probability in fission reactors. 
In one of the most remarkable phenomena in nature, a slow neutron can be captured by a uranium-235 nucleus, rendering it unstable toward nuclear fission. Other fissionable isotopes which can be induced to fission by slow neutrons are plutonium-239, uranium-233, and thorium-232. The fission of U-235 in reactors is triggered by the absorption of a low energy neutron, often termed a "slow neutron" or a "thermal neutron". For elements lighter than iron, fusion will yield energy. If the mass of the fragments is equal to or greater than that of iron at the peak of the binding energy curve, then the nuclear particles will be more tightly bound than they were in the uranium nucleus, and that decrease in mass comes off in the form of energy according to the Einstein equation. If a massive nucleus like uranium-235 breaks apart (fissions), then there will be a net yield of energy because the sum of the masses of the fragments will be less than the mass of the uranium nucleus. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
